In this study, we tested the hypothesis that specific and targeted inhibition of the NLRP3 inflammasome with MCC950 could protect against diabetes-associated atherosclerosis by reducing inflammation and improving vascular function. However, its efficacy has never been investigated in chronic inflammatory vascular disease induced by diabetes. Indeed, MCC950 was shown to attenuate carotid artery plaque size in a short-term model of atherosclerosis ( 22). One well-characterized NLRP3 small-molecule inhibitor is MCC950, which specifically binds to the NLRP3 ATP hydrolysis (or Walker B) motif, thereby preventing the second step in NLRP3 activation and IL-1β maturation ( 20, 21). Recent developments of specific NLRP3 small-molecule inhibitors have sparked great interest as anti-inflammatory therapeutics.
#Anova post hoc in graphpad prism 8 trial#
This pivotal trial provided the first proof-of-principle for the inflammation hypothesis in driving atherosclerosis in humans. The recent success of the CANTOS trial, a large-scale multicenter clinical trial that tested the efficacy of a monoclonal antibody (canakinumab) targeted at IL-1β, has solidified the clinical significance of targeting IL-1β to reduce cardiovascular events ( 1, 19). Moreover, in the clinical setting, increased expression of NLRP3, ASC, caspase-1, and IL-1β was observed in unstable carotid atherosclerotic plaques when compared with stable plaques and control patients who had no evidence of coronary artery stenosis ( 14). In diabetic mice, aortic protein expression of NLRP3, ASC, caspase-1, and IL-1β was upregulated compared with nondiabetic controls and was associated with enhanced lesion development and an elevation of reactive oxygen species levels ( 12), while knockdown of NLRP3 protected against diabetes-associated atherosclerosis ( 13). The importance of the NLRP3 inflammasome and its components in atherosclerotic disease is substantiated in several preclinical studies, which have demonstrated that deficiency in NLRP3, ASC, IL-1β, or caspase-1 protects against the development of diet-induced atherosclerosis ( 7, 10, 11). The fully formed NLRP3 inflammasome complex promotes caspase-1 cleavage and activation, which triggers the maturation of proinflammatory cytokines such as pro–interleukin-1β (IL-1β) and pro–IL-18 into their active secreted forms, triggering an inflammatory cascade ( 4, 9). In response to these damage-associated molecular patterns, NLRP3 binds to its adaptor protein, apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), which in turn recruits caspase-1.
#Anova post hoc in graphpad prism 8 free#
In the diabetic milieu, hyperglycemia and increased oxidative stress, together with elevated oxidized LDLs, free fatty acids, and cholesterol crystals, act as endogenous host-derived triggers for the assembly and activation of the NLRP3 inflammasome ( 7, 8). In summary, MCC950 reduces plaque development, promotes plaque stability, and improves vascular function, suggesting that targeting NLRP3-mediated inflammation is a novel therapeutic strategy to improve diabetes-associated vascular disease.

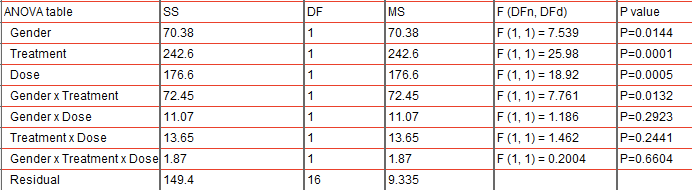
In a range of cell lines (murine bone marrow–derived macrophages, human monocytic THP-1 cells, phorbol 12-myristate 13-acetate–differentiated human macrophages, and aortic smooth muscle cells from humans with diabetes), MCC950 significantly reduced IL-1β and/or caspase-1 secretion and attenuated leukocyte–smooth muscle cell interactions under high glucose or lipopolysaccharide conditions. Additionally, vascular function was improved in diabetic vessels of mice treated with MCC950 ( P < 0.05). This reduction in lesions was associated with decreased monocyte–macrophage content, reduced necrotic core, attenuated inflammatory gene expression (IL-1β, tumor necrosis factor-α, intracellular adhesion molecule 1, and MCP-1 P < 0.05), and reduced oxidative stress, while maintaining fibrous cap thickness. Diabetes led to an approximately fourfold increase in atherosclerotic lesions throughout the aorta, which were significantly attenuated with MCC950 ( P < 0.001). We investigated whether inhibiting the NLRP3 inflammasome, through the use of the specific small-molecule NLRP3 inhibitor MCC950, could reduce inflammation, improve vascular function, and protect against diabetes-associated atherosclerosis in the streptozotocin-induced diabetic apolipoprotein E-knockout mouse. Low-grade persistent inflammation is a feature of diabetes-driven vascular complications, in particular activation of the Nod-like receptor family pyrin domain containing 3 (NLRP3) inflammasome to trigger the maturation and release of the inflammatory cytokine interleukin-1β (IL-1β).
